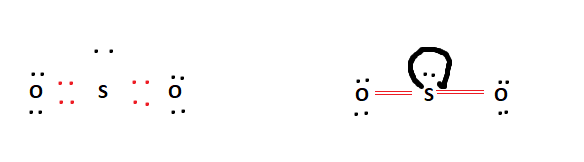
The 3d and 3p orbitals, on the other hand, stay unchanged and play a role in the creation of pi bonds. The oxygen atoms subsequently form sigma bonds with the unpaired electrons. The VSEPR notation of sulfur dioxide is AX 2 E 1. Both sulfur and oxygen are sp2 hybridized. Explain the principles of the VSEPR theory. Using VSEPR theory explain why 1)BF 4 + is a tetrahedral molecule 2)SF 3 + is a Trigonal pyramidal molecule 3)ICI 4 IS A SQUARE PLANER molecule 4)IF 5 IS A SQUARE pyramidal molecule 5)PF 5 Use VSEPR theory to predict the molecular geometry of H3O+. 2) SF3+ is a Trigonal pyramidal molecule. Use VSEPR theory to predict the shape and bond angle of SO2. The H-N-H bond angle is 109.5 degrees in the tetrahedral molecular geometry. Because the center atom, nitrogen, has four N-H bonds with the hydrogen atoms surrounding it. According to the VSEPR theory, the NH4+ molecule ion possesses tetrahedral molecular geometry. Using VSEPR THEORY explain why 1) BF4- is a tetrahedral molecule. Overview: NH4+ electron and molecular geometry. Explain the VSEPR theory in detail and give examples of molecules for each molecular shape. A quick explanation of the molecular geometry of SO3 2- (Sulfite ion) including a description of the SO3 2- bond angles.Looking at the SO3 2- Lewis structure. So let's just focus on one of these structures. Notice that you can draw resonance structures for SO2, but because VSEPR doesn't distinguish b/w single and multiple bonds, we can look at any resonance structure when predicting geometry. The exact shape of SO 2 is angular or bent with a bond angle of 119. Use the VSEPR theory to explain how BCl3 and NH3 differ in shape. Chapter Break Now let's take a look at SO2. Since one of these substituents is a lone pair, the shape will be distorted. Two hybrid orbitals will have unpaired electrons, while the lone pair will be in one hybrid orbital. These three groups are approximately 120 apart, corresponding to a trigonal planar geometry. The chemical molecule $S\] hybrid orbitals forming. Because the center atom, sulfur, has two S-O double bonds with the two. This reduces the energy of the molecule and enhances its stability, determining the molecular shape. According to the VSEPR theory, the SO2 molecule possesses linear molecular geometry. According to the VSEPR theory, the SO2 molecule possesses linear molecular geometry. 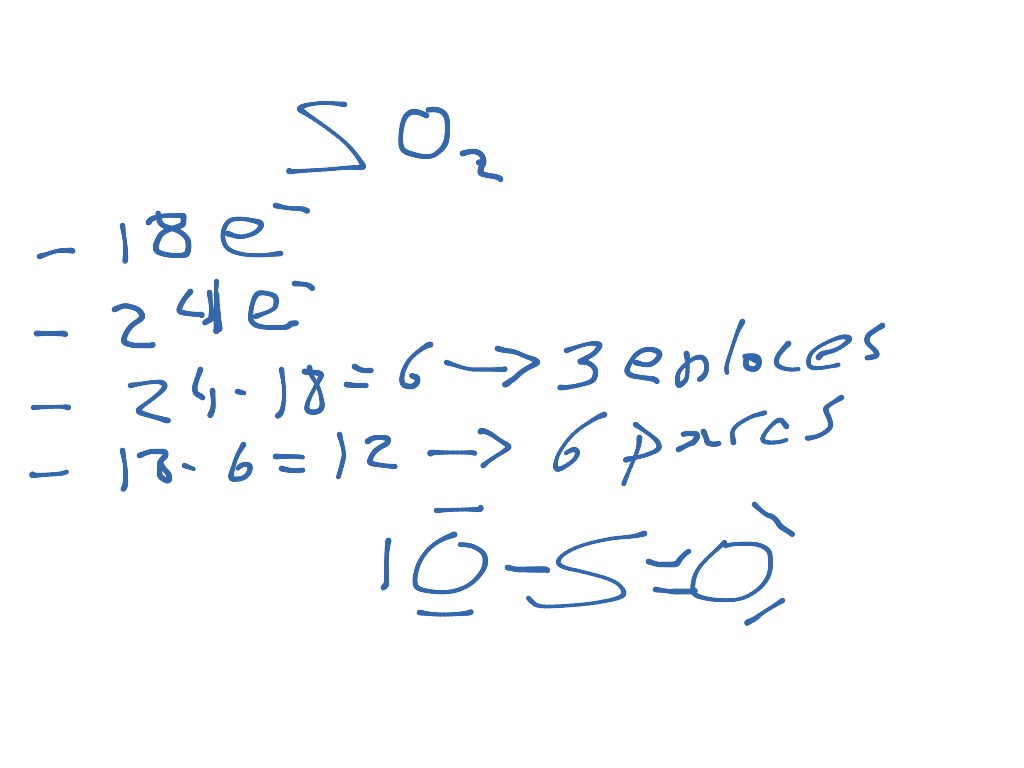
The assumption of VSEPR is that the valence electron pairs around an atom resist each other and, as a result, would organise themselves in a way that minimises this repulsion. Hint: The valence shell electron pair repulsion hypothesis is a chemical model that predicts a molecule's shape based on the number of electron pairs around its core atoms. The Valence-Shell-Electron-Pair-Repulsion (VSEPR) theory can predict molecular geometry by minimizing electron-electron repulsion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
